About NCVC
Cerebral small vessel disease: predictive for intracranial hemorrhage and probably for extracranial bleeding during antithrombotic therapy
Cerebral small vessel disease (SVD), which represents a risk factor for stroke, is one of the most prevalent neuropathological processes ever described. Cerebral SVD have received attention as a risk factor for antithrombotic-related intracranial hemorrhage. Researchers from the Bleeding with Antithrombotic Therapy (BAT) 2 study, a collaborative research at 52 hospitals in Japan, showed that cerebral SVD was not only a predictor intracranial hemorrhage but possibly also of extracranial bleeding among patients taking oral antithrombotic agents. The results suggest a broader clinical relevance for cerebral SVD as a marker for the safe implementation of antithrombotic therapy.
The team published their findings in the journal Annals of Neurology.
Antithrombotic agents (blood thinners), which prevent blood clots from forming, are widely used to prevent thromboembolic events such as ischemic stroke and myocardial infarction. This benefit of antithrombotic agents is not so rarely overshadowed by the drawback of an increased risk of bleeding complications, such as intracranial and gastrointestinal bleeding. Cerebral SVD can be a key finding in balancing the benefits (thromboembolism prevention) and disadvantages (bleeding complications) of antithrombotic therapy because it is common in elderly patients and those with hypertension and renal dysfunction, who are at high risk for stroke or cardiovascular disorders.
BAT2 is a multicenter prospective cohort study of patients with cerebrovascular and cardiovascular dieseases taking oral antithrombotic agents. Baseline multimodal MRI acquired under prespecified conditions were assessed by a central diagnostic radiology committee to evaluate cerebral SVD burden. The primary outcome was major bleeding, and secondary outcomes included bleeding at each site and ischemic events. The researchers analyzed data from BAT2 to determine the excess risk of antithrombotic-related bleeding due to cerebral SVD burden. The cerebral SVD burden was comprehensively quantified using total SVD score (range, 0–4), which incorporates the four typical MRI SVD markers of white matter hyperintentisy, lacunes, cerebral microbleeds, and enlarged basal ganglia perivascular spaces.
Of the analyzed 5,250 patients (9,933 patient-years of follow-up), the cumulative risk of major bleeding increased as the total SVD score increased from 0 to 4 (Figures A–C). The risk of ischemic events also increased significantly with increasing SVD score (Figure D). In patients with SVD score 4 compared to 0, the risks of major (adjusted hazard ratio [HR]: 5.47, 95% confidence interval [CI]: 2.26–13.23), intracranial (adjusted HR: 9.29, 95% CI: 1.99–43.35), extracranial major (adjusted HR: 3.43, 95% CI: 1.13–10.38), and gastrointestinal bleedings (adjusted HR: 2.54, 95% CI: 1.02–6.35) were 2.54- to 9.29-fold higher. The risk of ischemic event (adjusted HR: 1.76, 95% CI: 1.08–2.86) was 1.76-fold higher in SVD score 4 than in score 0.
Notably, in BAT2, event information was collected not only on cerebrovascular disease, but also on bleeding and ischemic events in other organs, allowing detailed observation of the relationships between cerebral SVD and antithrombotic-related outcome events. This feature of BAT2 led to find an association between total SVD score and extracranial major bleeding, most of which were due to gastrointestinal bleeding. Although further studies are needed before clear conclusions can be drawn regarding the association between cerebral SVD and extracranial bleeding, BAT2 suggest a novel clinical relevance of cerebral SVD beyond the previously demonstrated utility in stratifying the risk of antithrombotic-related intracranial hemorrhage.
Paper information
Authors: Kanta Tanaka, Kaori Miwa, Masatoshi Koga, Sohei Yoshimura, Kenji Kamiyama, Yoshiki Yagita, Yoshinari Nagakane, Haruhiko Hoshino, Tadashi Terasaki, Yasushi Okada, Yusuke Yakushiji, Shinichi Takahashi, Toshihiro Ueda, Yasuhiro Hasegawa, Masayuki Shiozawa, Makoto Sasaki, Kohsuke Kudo, Jun Tanaka, Masashi Nishihara, Yoshitaka Yamaguchi, Kyohei Fujita, Yuko Honda, Hiroyuki Kawano, Toshihiro Ide, Takeshi Yoshimoto, Masafumi Ihara, Teruyuki Hirano, Kazunori Toyoda, for BAT2 Investigators
Title: Cerebral small vessel disease burden for bleeding risk during antithrombotic therapy: Bleeding with Antithrombotic Therapy 2 study
Annals of Neurology
This work was supported by funding from
AMED under Grant Number, P18ek0210055, JP23lk0221171, and JP23lk0201109.
Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science under Grant Numbers, 19K17023.
Figure 1. Kaplan-Meier estimates for (A) major bleeding, (B) intracranial hemorrhage, (C) extracranial major bleeding, and (D) ischemic event by cerebral SVD burden
Cumulative risk of each outcome event was stratified by total SVD score.
Abbreviations: SVD, small vessel disease.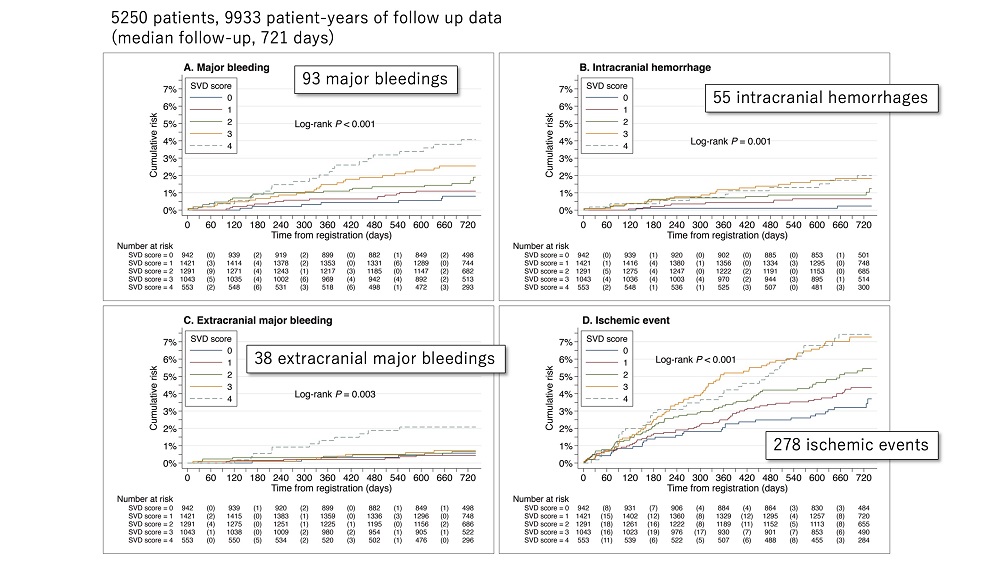
last updated:2023/12/28
